Oxidation of Alcohols
4. Oxidation Reactions of Alcohols
Simple 1º and 2º-alcohols in the gaseous state lose hydrogen when exposed to a hot copper surface. This catalytic dehydrogenation reaction produces aldehydes (as shown below) and ketones, and since the carbon atom bonded to the oxygen is oxidized, such alcohol to carbonyl conversions are generally referred to as oxidation reactions. Gas phase dehydrogenations of this kind are important in chemical manufacturing, but see little use in the research laboratory. Instead, alcohol oxidations are carried out in solution, using reactions in which the hydroxyl hydrogen is replaced by an atom or group that is readily eliminated together with the alpha-hydrogen. The decomposition of 1º and 2º-alkyl hypochlorites, referred to earlier, is an example of such a reaction.
RCH2–OH + hot Cu RCH=O + H2
RCH=O + H2
RCH2–O–Cl + base  RCH=O + H–Cl
RCH=O + H–Cl
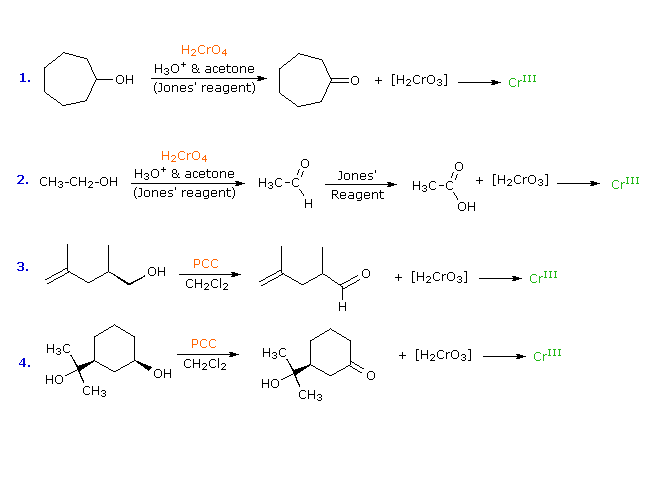
The most generally useful reagents for oxidizing 1º and 2º-alcohols are chromic acid derivatives. Two such oxidants are Jones reagent (a solution of sodium dichromate in aqueous sulfuric acid) and pyridinium chlorochromate, C5H5NH(+)CrO3Cl(–), commonly named by the acronym PCC and used in methylene chloride solution. In each case a chromate ester of the alcohol substrate is believed to be an intermediate, which undergoes an E2-like elimination to the carbonyl product. The oxidation state of carbon increases by 2, while the chromium decreases by 3 (it is reduced). Since chromate reagents are a dark orange-red color (VI oxidation state) and chromium III compounds are normally green, the progress of these oxidations is easily observed. Indeed, this is the chemical transformation on which the Breathalyzer test is based. The following equations illustrate some oxidations of alcohols, using the two reagents defined here. Both reagents effect the oxidation of 2º-alcohols to ketones, but the outcome of 1º-alcohol oxidations is different. Oxidation with the PCC reagent converts 1º-alcohols to aldehydes; whereas Jones reagent continues the oxidation to the carboxylic acid product, as shown in the second reaction. Reaction mechanisms for these transformations are displayed on clicking the "Show Mechanism" button. For the first two reactions the mechanism diagram also shows the oxidation states of carbon (blue Arabic numbers) and chromium (Roman numbers). The general base (B:) used in these mechanisms may be anything from water to pyridine, depending on the specific reaction.
Two structural requirements for the oxidation to
carbonyl products should now be obvious:
1. The carbon atom bonded to oxygen must also bear a hydrogen
atom.
Tertiary alcohols (R3C–OH) cannot be oxidized
in this fashion.
2. The oxygen atom must be bonded to a hydrogen
atom so that a chromate ester intermediate (or other suitable leaving
group) may be formed.
Ethers (R–O–R) cannot be oxidized in this
fashion.
The fourth reaction above illustrates the failure of 3º-alcohols to
undergo oxidation. The second reaction mechanism explains why
1º-alcohols undergo further oxidation by Jones reagent. The aqueous solvent
system used with this reagent permits hydration (addition of water) to the
aldehyde carbonyl group. The resulting hydrate (structure shown below the
aldehyde) meets both the requirements stated above, and is further oxidized
by the same chromate ester mechanism. Water is not present when the PCC
reagent is used, so the oxidation stops at the aldehyde stage.
Another chromate oxidizing agent, similar to PCC, is pyridinium dichromate,
(C5H5NH(+) )2
Cr2O7(–2), known by the acronym PDC. Both
PCC and PDC are orange crystalline solids that are soluble in many organic
solvents. Since PDC is less acidic than PCC it is often used to oxidize
alcohols that may be sensitive to acids. In methylene chloride solution,
PDC oxidizes 1º- and 2º-alcohols in roughly the same fashion as PCC, but
much more slowly. However, in DMF
solution saturated 1º-alcohols are oxidized to carboxylic acids. In both
solvents allylic alcohols are oxidized efficiently to conjugated enals and
enones respectively.
|
Other Reagents |
Phenols
Reactions of Phenols
Compounds in which a hydroxyl group is bonded to an aromatic ring are called phenols. The chemical behavior of phenols is different in some respects from that of the alcohols, so it is sensible to treat them as a similar but characteristically distinct group. A corresponding difference in reactivity was observed in comparing aryl halides, such as bromobenzene, with alkyl halides, such as butyl bromide and tert-butyl chloride. Thus, nucleophilic substitution and elimination reactions were common for alkyl halides, but rare with aryl halides. This distinction carries over when comparing alcohols and phenols, so for all practical purposes substitution and/or elimination of the phenolic hydroxyl group does not occur.
1. Acidity of Phenols
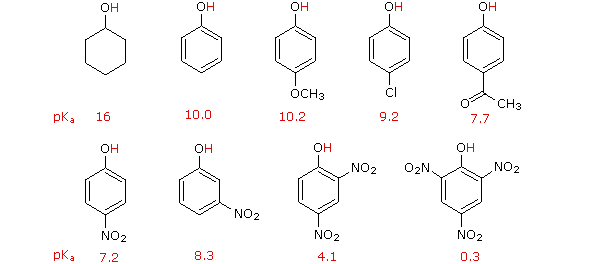
On the other hand, substitution of the hydroxyl hydrogen atom is even more facile with phenols, which are roughly a million times more acidic than equivalent alcohols. This phenolic acidity is further enhanced by electron-withdrawing substituents ortho and para to the hydroxyl group, as displayed in the following diagram. The alcohol cyclohexanol is shown for reference at the top left. It is noteworthy that the influence of a nitro substituent is over ten times stronger in the para-location than it is meta, despite the fact that the latter position is closer to the hydroxyl group. Furthermore additional nitro groups have an additive influence if they are positioned in ortho or para locations. The trinitro compound shown at the lower right is a very strong acid called picric acid.
Why is phenol a much stronger acid than cyclohexanol? To answer this
question we must evaluate the manner in which an oxygen substituent
interacts with the benzene ring. As noted in our earlier treatment of
electrophilic aromatic substitution reactions, an oxygen
substituent enhances the reactivity of the ring and favors electrophile
attack at ortho and para sites. It was proposed that resonance
delocalization of an oxygen non-bonded electron pair into the pi-electron
system of the aromatic ring was responsible for this substituent effect.
Formulas illustrating this electron delocalization will be displayed when
the "Resonance Structures" button beneath the previous diagram is clicked.
A similar set of resonance structures for the phenolate anion conjugate
base appears below the phenol structures. 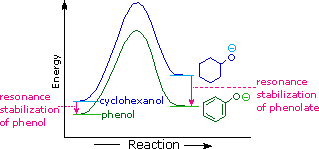
The resonance stabilization in these two cases is very different. An
important principle of
resonance is that charge separation diminishes the importance of
canonical contributors to the resonance hybrid and reduces the overall
stabilization. The contributing structures to the phenol hybrid all suffer
charge separation, resulting in very modest stabilization of this compound.
On the other hand, the phenolate anion is already charged, and the
canonical contributors act to disperse the charge, resulting in a
substantial stabilization of this species. The conjugate bases of simple
alcohols are not stabilized by charge delocalization, so the acidity of
these compounds is similar to that of water. An energy diagram showing the
effect of resonance on cyclohexanol and phenol acidities is shown on the
right. Since the resonance stabilization of the phenolate conjugate base is
much greater than the stabilization of phenol itself, the acidity of phenol
relative to cyclohexanol is increased. Supporting evidence that the
phenolate negative charge is delocalized on the ortho and para carbons of
the benzene ring comes from the influence of electron-withdrawing
substituents at those sites. The additional resonance stabilization
provided by ortho and para nitro substituents will be displayed by clicking
the "Resonance Structures" button a second time. You may cycle through
these illustrations by repeated clicking of the button.
2. Substitution of the Hydroxyl Hydrogen
As with the alcohols, the phenolic hydroxyl hydrogen is rather easily replaced by other substituents. For example, phenol reacts easily with acetic anhydride to give phenyl acetate. Likewise, the phenolate anion is an effective nucleophile in SN2 reactions, as in the second example below.
 C6H5–O–COCH3 +
CH3CO2H
C6H5–O–COCH3 +
CH3CO2H
C6H5–O(–) Na(+) +
CH3CH2CH3–Br  C6H5–O–CH2CH2CH3 +
NaBr
C6H5–O–CH2CH2CH3 +
NaBr
3. Electrophilic Substitution of the Phenol Aromatic Ring
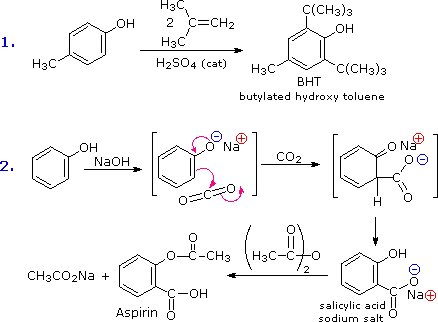
The facility with which the aromatic ring of phenols and phenol ethers undergoes electrophilic substitution has been noted. Two examples are shown in the following diagram. The first shows the Friedel-Crafts synthesis of the food preservative BHT from para-cresol. The second reaction is interesting in that it further demonstrates the delocalization of charge that occurs in the phenolate anion. Carbon dioxide is a weak electrophile and normally does not react with aromatic compounds; however, the negative charge concentration on the phenolate ring enables the carboxylation reaction shown in the second step. The sodium salt of salicylic acid is the major product, and the preference for ortho substitution may reflect the influence of the sodium cation. This is called the Kolbe-Schmidt reaction, and it has served in the preparation of aspirin, as the last step illustrates.
4. Oxidation of Phenols
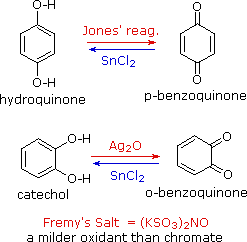
Phenols are rather easily oxidized despite the absence of a hydrogen
atom on the hydroxyl bearing carbon. Among the colored products from the
oxidation of phenol by chromic acid is the dicarbonyl compound
para-benzoquinone (also known as 1,4-benzoquinone or simply
quinone); an ortho isomer is also known. These compounds are easily reduced
to their dihydroxybenzene analogs, and it is from these compounds that
quinones are best prepared. Note that meta-quinones having similar
structures do not exist. The redox equilibria between the dihydroxybenzenes
hydroquinone and catechol and their quinone oxidation states are so facile
that milder oxidants than chromate (Jones reagent) are generally preferred.
One such oxidant is Fremy's salt, shown on the right. Reducing
agents other than stannous chloride (e.g. NaBH4) may be used for
the reverse reaction.
The position of the quinone-hydroquinone redox equilibrium is proportional
to the square of the hydrogen ion concentration, as shown by the following
half-reactions (electrons are colored blue). The electrode potential for
this interconversion may therefore be used to measure the pH of
solutions.
Quinone + 2H(+) |
2e(–) –2e(–) |
Hydroquinone |
|---|
Although chromic acid oxidation of phenols having an unsubstituted para-position gives some p-quinone product, the reaction is complex and is not synthetically useful. It has been found that salcomine, a cobalt complex, binds oxygen reversibly in solution, and catalyzes the oxidation of various substituted phenols to the corresponding p-quinones. The structure of salcomine and an example of this reaction are shown in the following equation. The solvent of choice for these oxidations is usually methanol or dimethylformamide (DMF).
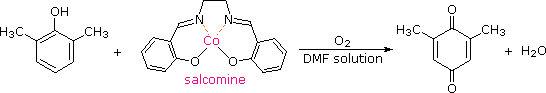
Practice Problems
|
The following problems review many aspects of alcohol and phenol chemistry. The first three questions concerns the nomenclature of alcohols. The fourth explores reactions of isobutanol. The fifth question focuses on the carbonyl products of many alcohol reactions. The sixth explores the many functional relationships of alcohols with other classes of compounds. The seventh asks you to draw the product of a reaction selected from 48 possible combinations of alcohols and reagents. |
Ethers
The Chemistry of Ethers
1. Nomenclature
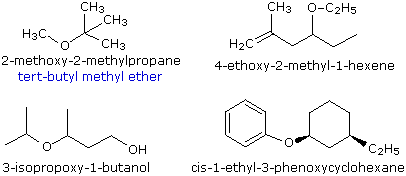
Ethers are compounds having two alkyl or aryl groups bonded to an oxygen atom, as in the formula R1–O–R2. The ether functional group does not have a characteristic IUPAC nomenclature suffix, so it is necessary to designate it as a substituent. To do so the common alkoxy substituents are given names derived from their alkyl component, as shown in the table on the right below. Examples of ether nomenclature are provided on the left. Simple ethers are given common names in which the alkyl groups bonded to the oxygen are named in alphabetical order followed by the word "ether". The top left example shows the common name in blue under the IUPAC name. Many simple ethers are symmetrical, in that the two alkyl substituents are the same. These are named as "dialkyl ethers". Examples are: CH3CH2OCH2CH3, diethyl ether (sometimes referred to as ether), and CH3OCH2CH2OCH3, ethylene glycol dimethyl ether (glyme).
 |
|
Sulfur analogs of ethers (R–S–R') are called sulfides. For example, (CH3)3C–S–CH3 is tert-butyl methyl sulfide. Sulfides are chemically more reactive than ethers, reflecting the greater nucleophilicity of sulfur relative to oxygen.
Ether Synthesis
2. Preparation of Ethers
Ethers are usually prepared from alcohols or their conjugate bases. One important procedure, known as the Williamson Ether Synthesis, proceeds by an SN2 reaction of an alkoxide nucleophile with an alkyl halide. Reactions #1 and #2 below are two examples of this procedure. When applied to an unsymmetrical ether, as in this case, there are two different combinations of reactants are possible. Of these one is usually better than the other. Since alkoxide anions are strong bases, the possibility of a competing E2 elimination must always be considered. Bearing in mind the factors that favor substitution over elimination, a 1º-alkyl halide should be selected as a preferred reactant whenever possible. Thus, reaction #1 gives a better and cleaner yield of benzyl isopropyl ether than does reaction #2, which generates considerable elimination product.
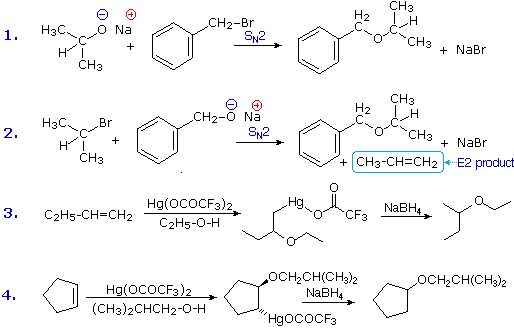
A second general ether synthesis, alkoxymercuration, is patterned after the oxymercuration reaction. Reactions #3 and #4 are examples of this two-step procedure. Note that the alcohol reactant is used as the solvent, and a trifluoroacetate mercury (II) salt is used in preference to the acetate (trifluoroacetate anion is a poorer nucleophile than acetate). The mechanism of alkoxymercuration is similar to that of oxymercuration, with an initial anti-addition of the mercuric species and alcohol being followed by reductive demercuration.
Acid-catalyzed dehydration of small 1º-alcohols constitutes a specialized method of preparing symmetrical ethers. As shown in the following two equations, the success of this procedure depends on the temperature. At 110º to 130 ºC an SN2 reaction of the alcohol conjugate acid leads to an ether product. At higher temperatures (over 150 ºC) an E2 elimination takes place.
| 2 CH3CH2-OH + H2SO4 | 130 ºC |
CH3CH2-O-CH2CH3 + H2O |
| CH3CH2-OH + H2SO4 | 150 ºC |
CH2=CH2 + H2O |
Reactions of Ethers
3. Reactions of Ethers
Ethers are widely used as solvents for a variety of organic compounds
and reactions, suggesting that they are relatively unreactive themselves.
Indeed, with the exception of the alkanes, cycloalkanes and fluorocarbons,
ethers are probably the least reactive, common class of organic compounds.
The inert nature of the ethers relative to the alcohols is undoubtedly due
to the absence of the reactive O–H bond.
The most common reaction of ethers is cleavage of the C–O bond by strong
acids. This may occur by SN1 or E1 mechanisms for 3º-alkyl
groups or by an SN2 mechanism for 1º-alkyl groups. Some examples
are shown in the following diagram. The conjugate acid of the ether is an
intermediate in all these reactions, just as conjugate acids were
intermediates in certain alcohol
reactions.
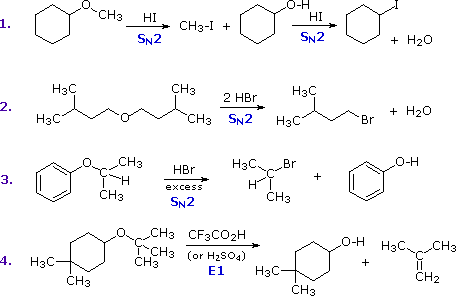
The first two reactions proceed by a sequence of SN2 steps in
which the iodide or bromide anion displaces an alcohol in the first step,
and then converts the conjugate acid of that alcohol to an alkyl halide in
the second. Since SN2 reactions are favored at least hindered
sites, the methyl group in example #1 is cleaved first. The 2º-alkyl group
in example #3 is probably cleaved by an SN2 mechanism, but the
SN1 alternative cannot be ruled out. The phenol formed in this
reaction does not react further, since SN2, SN1 and
E1 reactions do not take place on aromatic rings. The
last example shows the cleavage of a 3º-alkyl group by a strong acid. Acids
having poorly nucleophilic conjugate bases are often chosen for this
purpose so that E1 products are favored. The reaction shown here (#4) is
the reverse of the tert-butyl ether preparation described
earlier.
Ethers in which oxygen is bonded to 1º- and 2º-alkyl groups are subject to
peroxide formation in the presence of air (gaseous oxygen). This reaction
presents an additional hazard to the use of these flammable solvents, since
peroxides decompose explosively when heated or struck. The mechanism of
peroxide formation is believed to be free radical in nature (note that
molecular oxygen has two unpaired electrons).
| R–O–CH(CH3)2 + O2 |  |
R–O–C(CH3)2–O–O–H a peroxide |
|
Ethers as Protective Groups |
Epoxides
The Chemistry of Epoxides
Reactions of Epoxides
Epoxides (oxiranes) are three-membered cyclic ethers that are easily prepared from alkenes by reaction with peracids. Because of the large angle strain in this small ring, epoxides undergo acid and base-catalyzed C–O bond cleavage more easily than do larger ring ethers. Among the following examples, the first is unexceptional except for the fact that it occurs under milder conditions and more rapidly than other ether cleavages. The second and third examples clearly show the exceptional reactivity of epoxides, since unstrained ethers present in the same reactant or as solvent do not react. The aqueous acid used to work up the third reaction, following the Grignard reagent cleavage of the ethylene oxide, simply neutralizes the magnesium salt of the alcohol product.
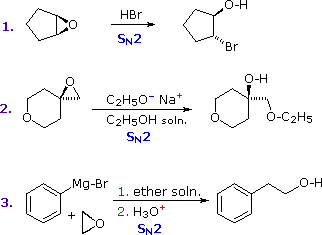
|
Sulfur Analogs of Alcohols and Ethers |
Practice Problems
|
Two problems involving the reactions of ethers are given here. |