Self-Replicating Cope Rearrangements
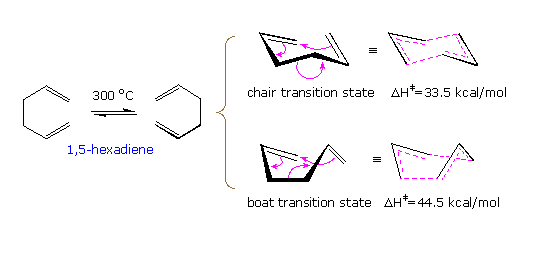
The simplest Cope rearrangement is the degenerate rearrangement of 1,5-hexatriene. As drawn in the following diagram, this [3.3]-sigmatropic shift may take place by one of two transition states, a lower energy chair state or a higher energy boat state. Because the product is identical with the reactant, isotopic labeling is needed to establish that change has occurred. The relatively high activation energy of this reaction requires a temperature near 300 ºC.
If ring strain is relieved in this rearrangement, as in the case of
cis-1,2 -divinylcyclopropane, the activation energy is substantially
reduced so that conversion to 1,4-cycloheptadiene occurs below 20 ºC within
30 minutes, as shown above by clicking on the
diagram. This rearrangement becomes degenerate for the compound
bicyclo[5.1.0]octa-2,7-diene, and the activation energy is further reduced.
In both cases a boat transition state is forced by structural
constraint.
If tricyclic analogs are formed by bridging the C1 and C8 carbons of
bicyclo[5.1.0]octa-2,7-diene, similar degenerate rearrangements are
observed. Examples will be displayed above by clicking
on the diagram again. For three of these compounds the activation
energy is even lower due to a combination of increased ground state strain
and transition state stabilization by interallylic bonding. The remarkable
C10H10 hydrocarbon bullvalene is exceptional in two
respects. First, its ground state is stabilized relative to the dihydro
analog by π-conjugation with the three-membered ring. Second, its
three-fold symmetry permits numerous self-replicating Cope rearrangements
leading to over a million (10!/3=1,209,600) identical valence tautomers.
All the carbons and hydrogens in bullvalene appear to be equivalent on the
nmr time scale at 100 ºC, and cannot be discriminated until cooled to -15
ºC. Molecules of this kind are termed fluxional. It is important to
recognize that valence tautomerization of this kind is not resonance
(the relative positions of the atoms undergo small changes).
Bullvalene
|
|---|