Peptides & Proteins
1. The Peptide Bond
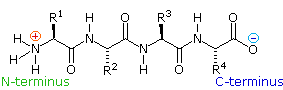
If the amine and carboxylic acid functional groups in amino acids join
together to form amide bonds, a chain of amino acid units, called a
peptide, is formed. A simple tetrapeptide structure is shown in the
following diagram. By convention, the amino acid component retaining a free
amine group is drawn at the left end (the N-terminus) of the peptide chain,
and the amino acid retaining a free carboxylic acid is drawn on the right
(the C-terminus). As expected, the free amine and carboxylic acid functions
on a peptide chain form a zwitterionic structure at their isoelectric
pH.
By clicking the "Grow Peptide" button, an
animation showing the assembly of this peptide will be displayed. The
"Show Structure" button displays some
bond angles and lengths that are characteristic of these compounds.
 |
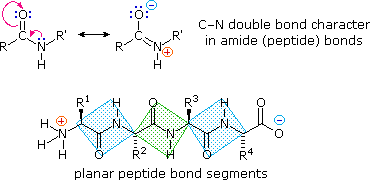
The conformational flexibility of peptide chains is limited chiefly to rotations about the bonds leading to the alpha-carbon atoms. This restriction is due to the rigid nature of the amide (peptide) bond. As shown in the following diagram, nitrogen electron pair delocalization into the carbonyl group results in significant double bond character between the carbonyl carbon and the nitrogen. This keeps the peptide links relatively planar and resistant to conformational change. The color shaded rectangles in the lower structure define these regions, and identify the relatively facile rotations that may take place where the corners meet (i.e. at the alpha-carbon). This aspect of peptide structure is an important factor influencing the conformations adopted by proteins and large peptides.
2. The Primary Structure of Peptides
Because the N-terminus of a peptide chain is distinct from the
C-terminus, a small peptide composed of different aminoacids may have a
several constitutional isomers. 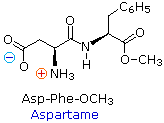 For example, a dipeptide
made from two different amino acids may have two different structures.
Thus, aspartic acid (Asp) and phenylalanine (Phe) may be combined to make
Asp-Phe or Phe-Asp, remember that the amino acid on the left is the
N-terminus. The methyl ester of the first dipeptide (structure on the
right) is the artificial sweetener aspartame, which is nearly 200
times sweeter than sucrose. Neither of the component amino acids is sweet
(Phe is actually bitter), and derivatives of the other dipeptide (Phe-Asp)
are not sweet.
For example, a dipeptide
made from two different amino acids may have two different structures.
Thus, aspartic acid (Asp) and phenylalanine (Phe) may be combined to make
Asp-Phe or Phe-Asp, remember that the amino acid on the left is the
N-terminus. The methyl ester of the first dipeptide (structure on the
right) is the artificial sweetener aspartame, which is nearly 200
times sweeter than sucrose. Neither of the component amino acids is sweet
(Phe is actually bitter), and derivatives of the other dipeptide (Phe-Asp)
are not sweet.
A tripeptide composed of three different amino acids can be made in 6
different constitutions, and the tetrapeptide shown above (composed of four
different amino acids) would have 24 constitutional isomers. When all
twenty of the natural amino acids are possible components of a peptide, the
possible combinations are enormous. Simple statistical probability
indicates that the decapeptides made up from all possible combinations of
these amino acids would total 2010!
Natural peptides of varying complexity are abundant. The simple and
widely distributed tripeptide glutathione (first entry in the following
table), is interesting because the side-chain carboxyl function of the
N-terminal glutamic acid is used for the peptide bond. An N-terminal
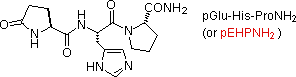
glutamic acid may also close to a lactam ring, as in the case of TRH
(second entry). The abbreviation for this transformed unit is pGlu (or pE),
where p stands for "pyro" (such ring closures often occur on heating). The
larger peptides in the table also demonstrate the importance of amino acid
abbreviations, since a full structural formula for a nonapeptide (or
larger) would prove to be complex and unwieldy. The formulas using single
letter abbreviations are colored red.
The ten peptides listed in this table make use of all twenty common amino
acids. Note that the C-terminal unit has the form of an amide in some cases
(e.g. TRH, angiotensin & oxytocin). When two or more cysteines are
present in a peptide chain, they are often joined by disulfide bonds
(e.g. oxytocin & endothelin); and in the case of insulin, two separate
peptide chains (A & B) are held together by such links.
Some Common Natural Peptides
|
Name (residues) |
Source or Function |
Amino Acid Sequence |
|---|---|---|
| Glutathione (3) | Most Living Cells (stimulates tissue growth) |
(+)H3NCH(CO2(–))CH2CH2CONHCH(CH2SH)CONHCH2CO2H γ-Glu-Cys-Gly (or γECG) |
| TRH (3) | Hypothalmic Neurohormone (governs release of thyrotropin) |
 |
| Angiotensin II (8) | Pressor Agent (acts on the adrenal gland) |
Asp-Arg-Val-Tyr-Ile-His-Pro-PheNH2 (or DRVYIHPFNH2) |
| Bradykinen (9) | Hypotensive Vasodilator (acts on smooth muscle) |
Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg (or RPPGFSPFR) |
| Oxytocin (9) | Uterus-Contracting Hormone (also stimulates lactation) |
 |
| Somatostatin (14) | Inhibits Growth Hormone Release (used to treat ulcers) |
 |
| Endothelin (21) | Potent Vasoconstrictor (structurally similar to some snake venoms) |
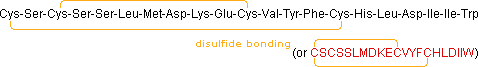 |
| Melittin (26) | Honey Bee Venom (used to treat rheumatism) |
Gly-Ile-Gly-Ala-Val-Leu-Lys-Val-Leu-Thr-Thr-Gly-Leu-Pro~ ~Ala-Leu-Ile-Ser-Trp-Ile-Lys-Arg-Lys-Arg-Gln-GlnNH2 (or GIGAVLKVLTTGLPALISWIKRKRQQNH2) |
| Glucagon (29) | Hyperglycemic Factor (used as an anti-diabetic) |
His-Ser-Gln-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp~ ~Ser-Arg-Arg-Ala-Gln-Asp-Phe-Val-Gln-Trp-Leu-Met-Asn-Thr (or HSQGTFTSDYSKYLDSRRAQDFVQWLMNT) |
| Insulin (51) | Pancreatic Hormone (used in treatment of diabetes) |
 |
The different amino acids that make up a peptide or protein, and the
order in which they are joined together by peptide bonds is referred to as
the primary structure. From the examples shown above, it should be
evident that it is not a trivial task to determine the primary structure of
such compounds, even modestly sized ones.
Complete hydrolysis of a protein or peptide, followed by amino acid
analysis establishes its gross composition, but does not provide any
bonding sequence information.
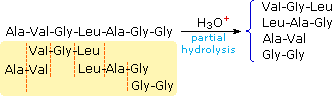
Partial hydrolysis will produce a mixture of shorter peptides and some amino acids. If the primary structures of these fragments are known, it is sometimes possible to deduce part or all of the original structure by taking advantage of overlapping pieces. For example, if a heptapeptide was composed of three glycines, two alanines, a leucine and a valine, many possible primary structures could be written. On the other hand, if partial hydrolysis gave two known tripeptide and two known dipeptide fragments, as shown on the right, simple analysis of the overlapping units identifies the original primary structure. Of course, this kind of structure determination is very inefficient and unreliable. First, we need to know the structures of all the overlapping fragments. Second, larger peptides would give complex mixtures which would have to be separated and painstakingly examined to find suitable pieces for overlapping. It should be noted, however, that modern mass spectrometry uses this overlap technique effectively. The difference is that bond cleavage is not achieved by hydrolysis, and computers assume the time consuming task of comparing a multitude of fragments.
3. N-Terminal Group Analysis
Over the years that chemists have been studying these important natural
products, many techniques have been used to investigate their primary
structure or amino acid sequence. Indeed, commercial instruments that
automatically sequence peptides and proteins are now available. A few of
the most important and commonly used techniques will be described here.
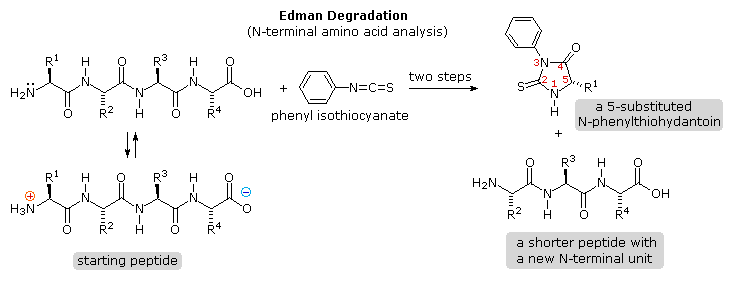
Identification of the N-terminal and C-terminal aminoacid units of a
peptide chain provides helpful information. N-terminal analysis is
accomplished by the Edman Degradation, which is outlined in the
following diagram. A free amine function, usually in equilibrium with
zwitterion species, is necessary for the initial bonding to the phenyl
isothiocyanate reagent. The products of the Edman degradation are a
thiohydantoin heterocycle incorporating the N-terminal amino acid together
with a shortened peptide chain. Amine functions on a side-chain, as in
lysine, may react with the isothiocyanate reagent, but do not give
thiohydantoin products.
 |
Repeated clicking of the "Next
Diagram" button displays the mechanism of this important
analytical method.
A major advantage of the Edman procedure is that the remaining peptide
chain is not further degraded by the reaction. This means that the
N-terminal analysis may be repeated several times, thus providing the
sequence of the first three to five amino acids in the chain. A
disadvantage of the procedure is that is peptides larger than 30 to 40
units do not give reliable results.
4. C-Terminal Group Analysis
Chemical Analysis
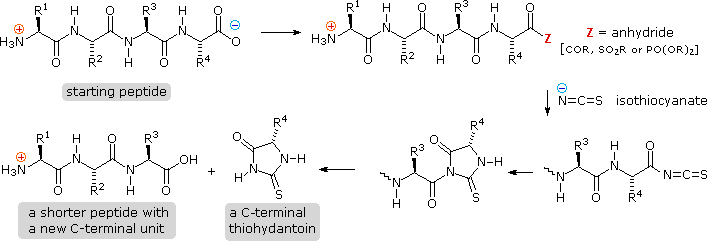
Complementary C-terminal analysis of peptide chains may be accomplished
chemically or enzymatically. The chemical analysis is slightly more complex
than the Edman procedure. First, side-chain carboxyl groups and hydroxyl
groups must be protected as amides or esters. Next, the C-terminal carboxyl
group is activated as an anhydride and reacted with thiocyanate. The
resulting acyl thiocyanate immediately cyclizes to a hydantoin ring, and
this can be cleaved from the peptide chain in several ways, not described
here. Depending on the nature of this final cleavage, the procedure can be
modified to give a C-terminal acyl thiocyanate peptide product which
automatically rearranges to a thiohydantoin incorporating the penultimate
C-terminal unit. Thus, repetitive analyses may be conducted in much the
same way they are with the Edman procedure.
Enzymatic Analysis
Enzymatic C-terminal amino acid cleavage by one of several carboxypeptidase
enzymes is a fast and convenient method of analysis. Because the shortened
peptide product is also subject to enzymatic cleavage, care must be taken
to control the conditions of reaction so that the products of successive
cleavages are properly monitored. The following example illustrates this
feature. A peptide having a C-terminal sequence: ~Gly-Ser-Leu is subjected
to carboxypeptidase cleavage, and the free aminoacids cleaved in this
reaction are analyzed at increasing time intervals. By
clicking on the diagram, the results of this experiment will be
displayed. The leucine is cleaved first, the serine second, and the glycine
third, as demonstrated by the sequential analysis. Of course, fourth and
fifth units will also be released as time passes, but these products are
not shown.
5. Selective Peptide Cleavage
|
Mechanisms for the enzymatic reactions are not as easily formulated. Other enzymatic cleavages have been developed, but the two listed here will serve to illustrate their application.
An Example of Primary Structure Analysis
To see how these procedures can be combined to elucidate the primary structure of a peptide, consider the melanocyte stimulating hormone isolated from pigs. This octadecapeptide (18 amino acid units) has the composition: Arg,Asp2,Glu2,Gly2,His,Lys2,Met,Phe,Pro3,Ser,Tyr2, and is abbreviated P18. The following diagram, which begins with the results of terminal unit analysis, illustrates the logical steps that could be used to solve the structural problem. By clicking the "Next Stage" button the results and conclusions from each step will be displayed. Comments about each stage are presented under the diagram.
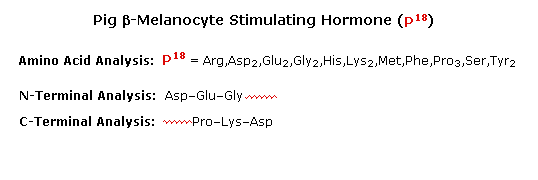 |
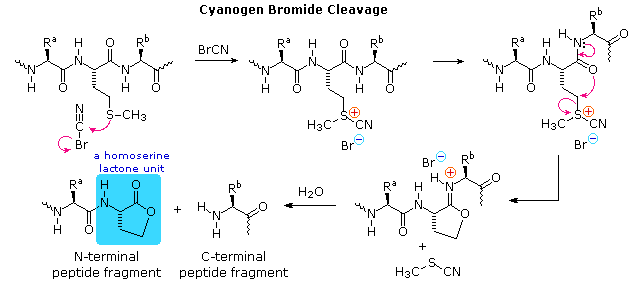
a) Cyanogen bromide cleavage gives two peptide fragments, the
longer of which has all the units on the C-terminal side of
methionine.
b) N-terminal analysis of the undecapeptide fragment,
P11, locates the three amino acids to the right of
methionine.
c) Trypsin cleavage of P11 shows the location of
the single arginine, which is found as the C-terminal unit of the
tetrapeptide fragment. One of the two lysines was known to be next to the
C-terminus. The other must be part of the smaller peptide from the
cyanogen bromide reaction.
d) With only four amino acids remaining to be located, the
position of the second tyrosine may be pursued by chymotrypsin cleavage
of P18 itself. Four fragments are obtained, and the
final structure might have been solved by these alone. However, selective
terminal group analysis of the two pentapeptides serves to locate the
tyrosine and a second proline next to the left most glycine, as well as
identifying the units on each side of the methionine. The one remaining
amino acid, a proline, is then placed at the last vacant site (yellow
box).
6. Cyclic Peptides
If the carboxyl function at the C-terminus of a peptide forms a peptide bond with the N-terminal amine group a cyclic peptide is formed. Carboxyate and amine functions on side chains may also combine to form rings. Cyclic peptides are most commonly found in microorganisms, and often incorporate some D-amino acids as well as unusual amino acids such as ornithine (Orn). The decapeptide antibiotic gramacidin S, produced by a strain of Bacillus brevis, is one example of this interesting class of natural products. The structure of gramicidin S is shown in the following diagram. The atypical amino acids are colored. When using a shorthand notation for cyclic structures, the top line is written by the usual convention (N-group on the left), but vertical and lower lines must be adjusted to fit the bonding. Arrows on these bonds point in the CO-N direction of each peptide bond.
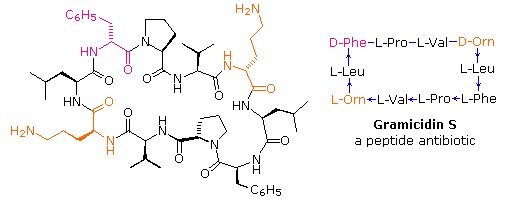
To see a model of another cyclic peptide, having potentially useful medicinal properties Click Here.
Structure-Property Relationships
The compounds we call proteins exhibit a broad range of physical and biological properties. Two general categories of simple proteins are commonly recognized.
| Fibrous Proteins | As the name implies, these substances have fiber-like structures,
and serve as the chief structural material in various tissues.
Corresponding to this structural function, they are relatively
insoluble in water and unaffected by moderate changes in temperature
and pH. Subgroups within this category include: Collagens & Elastins, the proteins of connective tissues. tendons and ligaments. Keratins, proteins that are major components of skin, hair, feathers and horn. Fibrin, a protein formed when blood clots. |
|
|---|---|---|
| Globular Proteins | Members of this class serve regulatory, maintenance and catalytic
roles in living organisms. They include hormones, antibodies and enzymes. and either dissolve or form colloidal suspensions in water. Such proteins are generally more sensitive to temperature and pH change than their fibrous counterparts. More Information |
1. The Secondary and Tertiary Structure of Large Peptides and Proteins
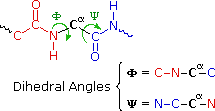
The various properties of peptides and proteins depend not only on their component amino acids and their bonding sequence in peptide chains, but also on the way in which the peptide chains are stretched, coiled and folded in space. Because of their size, the orientational options open to these macromolecules might seem nearly infinite. Fortunately, several factors act to narrow the structural options, and it is possible to identify some common structural themes or secondary structures that appear repeatedly in different molecules. These conformational segments are sometimes described by the dihedral angles Φ & Ψ, defined in the diagram on the right below. Most proteins and large peptides do not adopt completely uniform conformations, and full descriptions of their preferred three dimensional arrangements are defined as tertiary structures.
|
Five factors that influence the
conformational equilibria of peptide chains are: |
 |
A. Helical Coiling
The relatively simple undecapeptide shown in the following diagram can
adopt a zig-zag linear conformation, as drawn. A ball & stick model of
this peptide will be displayed by clicking the appropriate button. However,
this molecule prefers to assume a coiled helical conformation, displayed by
clicking any of the three buttons on the right. The middle button shows a
stick model of this helix, with the backbone chain drawn as a heavy black
line and the hydrogen bonds as dashed maroon lines. The other buttons
display a ball & stick model and a ribbon that defines this
α-helix. Seven hydrogen bonds, that together provide roughly 30
kcal/mol stability, help to maintain this conformation.
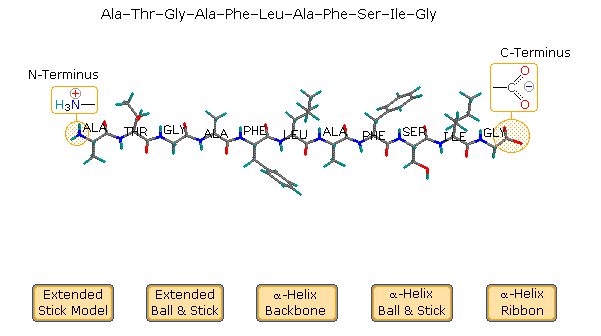
Examine the drawing activated by the middle button. The N-terminal
residue (Ala) is on the left, and the C-terminal Gly on the right. The
alpha-helix is right-handed, which means that it rotates clockwise as it
spirals away from a viewer at either end. Other structural features that
define an alpha-helix are: the relative locations of the donor and acceptor
atoms of the hydrogen bond, the number of amino acid units per helical turn
and the distance the turn occupies along the helical axis. The first
hydrogen bond (from the N-terminal end) is from the carbonyl group of the
alanine to the N-H group of the phenylalanine. Three amino acids, Thr, Gly
& Ala, fall entirely within this turn. Parts of the N-terminal alanine
acceptor and the phenylalanine donor also fall within this helical turn,
and careful analysis of the structure indicates there are 3.6 amino acid
units per turn. The distance covered by the turn is 5.4 Å. Using the
dihedral angle terminology noted above, a perfect α-helix has Φ = -58º and
Ψ = -47º. In natural proteins the values associated with α-helical
conformations range from -57 to -70º for Φ, and from -35 to -48º for Ψ. To
examine a model of this alpha-helix, click on the green circle. Once this
display is activated, the important hormone insulin may be shown by
clicking the appropriate button in the blue-shaded rows.
Helical conformations of peptide chains may also be described by a two
number term, nm, where n is the number of amino
acid units per turn and m is the number of atoms in the smallest
ring defined by the hydrogen bond. Using this terminology, the alpha-helix
is a 3.613 helix. Other common helical conformations are
310 and 4.416. The alpha helix is the most stable of
these, accounting for a third of the secondary structure found in most
globular (non-fibrous) proteins.
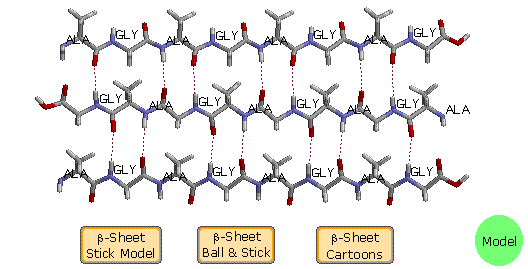
B. β-Pleated Sheets
The linear zig-zag conformation of a peptide chain may be stabilized by
hydrogen bonding to adjacent parallel chains of the same kind. Bulky
side-chain substituents destabilize this arrangement due to steric
crowding, so this beta-sheet conformation is usually limited to
peptides having a large amount of glycine and alanine. Steric interactions
also cause a slight bending or contraction of the peptide chains, and this
results in a puckered distortion (the pleated sheet). As shown in the
following diagram, the adjacent chains may be oriented in opposite N to C
directions, termed antiparallel. Using the dihedral angle
terminology, an antiparallel β-sheet has Φ = -139º and a Ψ = 135º.
Alternatively, the adjacent peptide chains may be oriented in the same
direction, termed parallel. By convention, beta-sheets are
designated by broad arrows or cartoons, pointing in the direction of the
C-terminus. In this diagram, these cartoons (colored violet) are displayed
by clicking on the appropriate button. A model of a two-antiparallel-chain
structure may be examined by clicking on the green circle.
Some proteins have layered stacks of β-sheets, which impart structural
integrity and may open to form a cavity (a beta barrel). An example is
human retinol binding protein, which has a cavity formed by eight β-sheet
strands. A model of this interesting protein may be displayed by clicking
the upper button in the blue-shaded rows.
When beta-sheets are observed as secondary structural components of
globular proteins, they are twisted by about 5 to 25º per residue;
consequently, the planes of the sheets are not parallel. The twist is
always of the same handedness, and is usually greater for antiparallel
sheets. Examples will be found in the following structures.
C. Other Structures
Although most proteins and large peptides may have alpha-helix and
beta-sheet segments, their tertiary structures may consist of less highly
organized turns, strands and coils. Turns reverse the direction of
the peptide chain, and are considered to be a third common secondary
structure motif. Approximately a third of all the residues in globular
proteins are found in turns. Turns occur chiefly on the protein surface,
often incorporate polar and charged residues, and have been classified in
three sub-groups.
As noted earlier, several factors perturb the
organization of peptide chains. One that has not yet been cited is the
structural influence of proline. Unlike the other common amino acids,
rotation about the α C-N bond in proline is not possible due to the
structural constraint of the five-membered ring. Consequently, the presence
of a proline in a peptide chain introduces a bend or kink that disrupts
helices or sheets. Also, prolines that are part of a peptide chain have no
N-H hydrogen bonding donors to contribute to conformer stabilization.
With the exception of silk fibroin and certain synthetically engineered
peptides, significant portions of most proteins adopt conformations that
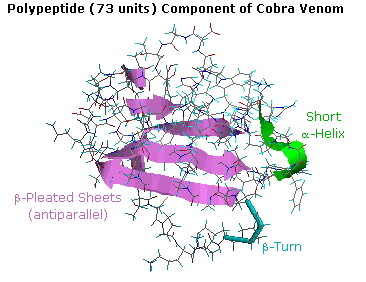
resist simple description or categorization. For example, the following
diagram shows the tertiary structure of a polypeptide neurotoxin found in
cobra venom. A large section of antiparallel beta-sheets is colored violet,
and a short alpha-helix is green. The remaining peptide chain seems
disorganized, but certain features such as a 180º turn (called a beta-turn)
and five disulfide bonds can be identified. A Chime model of this compound
may be examined by clicking on the
diagram.
Additional Examples
A full description and discussion of protein structure is beyond the
scope of this text, but a few additional examples will be instructive. In
addition to the tertiary structures that will be displayed, attention must
also be given to the way in which peptide structures may aggregate to form
dimeric, trimeric and tetrameric clusters. These assemblies, known as
quaternary structures, have characteristic properties different from
their monomeric components. The examples of mellitin, collagen and
hemoglobin, shown below demonstrate this feature.
Some proteins incorporate nonpeptide molecules in their overall structure,
either bonded covalently or positioned by other forces. These are called
conjugated proteins, and the non-peptide components are referred to
as prosthetic groups. Examples of conjugated proteins include:
Glycoproteins, incorporating polysaccharide prosthetic groups
(e.g. collagen and mucus).
Lipoproteins, incorporating lipid prosthetic groups (e.g. HDL and
LDL).
Chromoproteins, incorporating colored prosthetic groups (e.g.
hemoglobin).
The seven illustrations shown below identify a set of peptides and proteins that may be examined as Jmol models by clicking on a selected picture.
Endothelin & Angiogenin are small peptides that have
important and selective physiological properties.
Lysozyme a typical globular protein, incorporating many
identifiable secondary structures.
Mellitin, from honey bee venom, has a well-defined quaternary
structure, half of which is shown here.
Collagen is a widely distributed fibrous protein with a large and
complex quaternary structure. Only a small model segment is shown
here.
Thioredoxin is a relatively small regulatory protein serving an
important redox function.
Hemoglobin, the most complex of these examples, is a large
conjugated protein that transports oxygen. A 170 pound human has about a
kilogram of hemoglobin distributed among some five billion red blood
cells. A liter of arterial blood at body temperature can transport over
200 mL of oxygen, whereas the same fluid stripped of its hemoglobin will
carry only 2 to 3 mL. The supramolecular assembly of four subunits
exemplifies a quaternary structure.
2. Quaternary Structures of Proteins
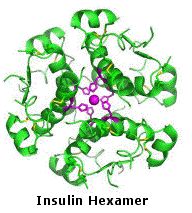
Many proteins are actually assemblies of several polypeptides, which in the context of the larger aggregate are known as protein subunits. Such multiple-subunit proteins possess a quaternary structure, in addition to the tertiary structure of the subunits. The subunits of a quaternary structure are held together by the same forces that are responsible for tertiary structure stabilization. These include hydrophobic attraction of nonpolar side chains in contact regions of the subunits, electrostatic interactions between ionic groups of opposite charge: hydrogen bonds between polar groups; and disulfide bonds. Examples of proteins having a quaternary (or quartary) structure include hemoglobin, HIV-1 protease and the insulin hexamer.
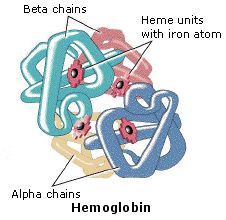 The hemoglobin
molecule is an assembly of four protein subunits, two alpha units and two
beta units. Each protein chain folds into a set of alpha-helix structural
segments connected together in a globin arrangement, so called because this
arrangement is the same folding motif used in other heme/globin proteins
such as myoglobin. This folding pattern contains a pocket which strongly
binds the heme group. The four polypeptide chains are bound to each other
by salt bridges, forming a tetrameric quaternary structure. A model of
hemoglobin was shown above, and may also be examined by clicking the image
on the left.
The hemoglobin
molecule is an assembly of four protein subunits, two alpha units and two
beta units. Each protein chain folds into a set of alpha-helix structural
segments connected together in a globin arrangement, so called because this
arrangement is the same folding motif used in other heme/globin proteins
such as myoglobin. This folding pattern contains a pocket which strongly
binds the heme group. The four polypeptide chains are bound to each other
by salt bridges, forming a tetrameric quaternary structure. A model of
hemoglobin was shown above, and may also be examined by clicking the image
on the left.
In animals, hemoglobin transports oxygen from the lungs or gills to the
rest of the body, where it releases the oxygen for cell use. Hemoglobin's
oxygen-binding capacity is decreased in the presence of carbon monoxide
because both gases compete for the same binding sites on hemoglobin. The
binding affinity of hemoglobin for CO is 200 times greater than its
affinity for oxygen. When hemoglobin combines with CO, it forms a very
bright red compound called carboxyhemoglobin, which may cause the skin of
CO poisoning victims to appear pink in death. In heavy smokers, up to 20%
of the oxygen-active sites can be blocked by CO. Similarly, hemoglobin has
a competitive binding affinity for cyanide, sulfur monoxide, nitrogen
dioxide and sulfides including hydrogen sulfide . All of these bind to the
heme iron without changing its oxidation state, causing grave
toxicity.
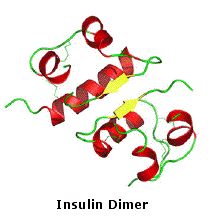
The molecular structure of insulin varies slightly between species of animals. Porcine (pig) insulin is especially close to the human version. Insulin molecules have a tendency to form dimers in solution due to hydrogen-bonding between the C-termini of B chains. In the presence of zinc ions, insulin dimers associate into hexamers. Insulin is stored in the body as a hexamer, whereas the active form is the monomer. These interactions have important clinical ramifications. Monomers and dimers readily diffuse into blood; hexamers diffuse poorly. By clicking the image on the far left, a model of the insulin monomer will be displayed . A model of the hexamer will be shown by clicking its image.
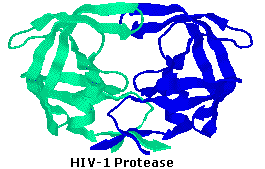 HIV-1 protease is an
enzyme made by the HIV virus that is essential for it's life-cycle. The
virus makes certain proteins that need to be cleaved or cut, in order to
transform into functional proteins that enable the virus to infect new
cells. HIV-1 protease cleaves the nascent proteins into their functional
form. The enzyme is composed of two symmetrically related subunits, shown
here in cartoon backbone representation to highlight the secondary
structure. Each subunit consists of the same small chain of 99 amino acids,
which come together in such as way as to form a tunnel where they meet. The
protein to be cleaved sits in this tunnel, which houses the active site of
the enzyme. Two Asp-Thr-Gly catalytic triads, one on each chain, compose
the active site. The two Asp's act as the main catalytic agents, and
together with a water molecule cleave the protein chain bound in the
tunnel. Without effective HIV-1 protease, HIV virions remain
uninfectious.
HIV-1 protease is an
enzyme made by the HIV virus that is essential for it's life-cycle. The
virus makes certain proteins that need to be cleaved or cut, in order to
transform into functional proteins that enable the virus to infect new
cells. HIV-1 protease cleaves the nascent proteins into their functional
form. The enzyme is composed of two symmetrically related subunits, shown
here in cartoon backbone representation to highlight the secondary
structure. Each subunit consists of the same small chain of 99 amino acids,
which come together in such as way as to form a tunnel where they meet. The
protein to be cleaved sits in this tunnel, which houses the active site of
the enzyme. Two Asp-Thr-Gly catalytic triads, one on each chain, compose
the active site. The two Asp's act as the main catalytic agents, and
together with a water molecule cleave the protein chain bound in the
tunnel. Without effective HIV-1 protease, HIV virions remain
uninfectious.Because of its role in HIV replication, HIV-1 protease has been a target for antiviral drugs. Such drugs function as inhibitors, binding to the active site by mimicking the tetrahedral intermediate of its substrate, thus disabling the enzyme. The structure of one such inhibitor, BEA388, will be displayed on the left by clicking here.
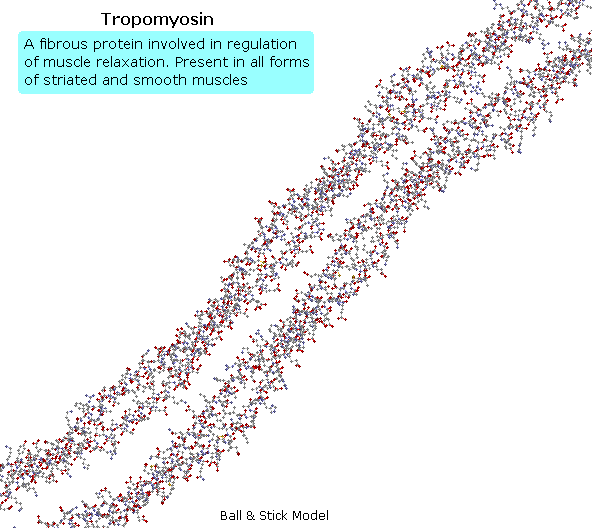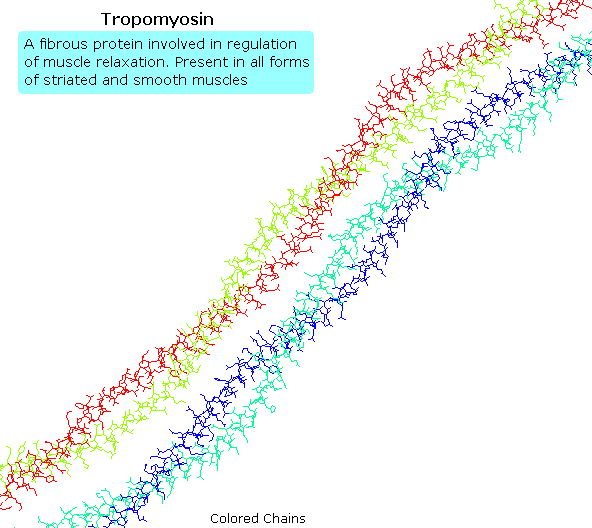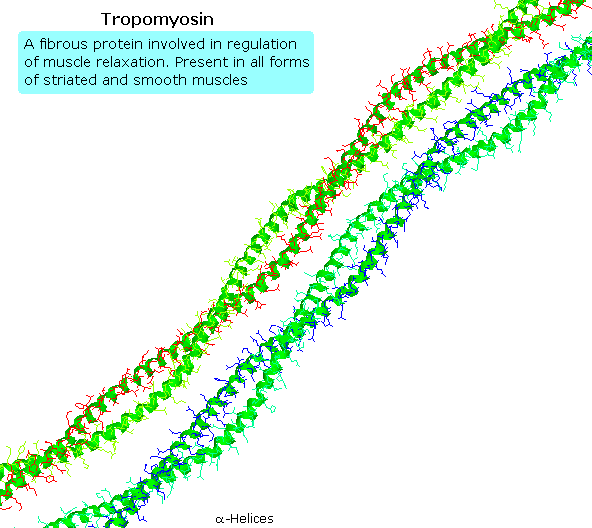
Tropomyosin
The following animation shows a segment of the fibrous protein tropomyosin, a common muscle regulator. The peptide chains are largely alpha-helices. These are wrapped in superhelix pairs, which are then aligned in a parallel array.If animation is not occurring, click on the drawing or reload.
| Two excellent resources of additional information are: First Glance Motivated Proteins Also, The Protein Data Bank provides a large collection of protein structures obtained by Xray and NMR. |
Peptide Synthesis
In order to synthesize a peptide from its component amino acids, two
obstacles must be overcome. The first of these is statistical in nature,
and is illustrated by considering the dipeptide Ala-Gly as a proposed
target. If we ignore the chemistry involved, a mixture of equal molar
amounts of alanine and glycine would generate four different dipeptides.
These are: Ala-Ala, Gly-Gly, Ala-Gly & Gly-Ala. In the case of
tripeptides, the number of possible products from these two amino acids
rises to eight. Clearly, some kind of selectivity must be exercised if
complex mixtures are to be avoided.
The second difficulty arises from the fact that carboxylic acids and 1º or
2º-amines do not form amide bonds on mixing, but will generally react by
proton transfer to give salts (the intermolecular equivalent of zwitterion
formation).
From the perspective of an organic chemist, peptide synthesis requires
selective acylation of a free amine. To accomplish the desired amide bond
formation, we must first deactivate all extraneous amine functions so they
do not compete for the acylation reagent. Then we must selectively activate
the designated carboxyl function so that it will acylate the one remaining
free amine. Fortunately, chemical reactions that permit us to accomplish
these selections are well known.
First, the basicity and nucleophilicity of amines are substantially
reduced by amide formation. Consequently, the acylation of amino acids
by treatment with acyl chlorides or anhydrides at pH > 10, as described earlier,
serves to protect their amino groups from further reaction.
Second, acyl halide or anhydride-like activation of a specific carboxyl
reactant must occur as a prelude to peptide (amide) bond formation. This is
possible, provided competing reactions involving other carboxyl functions
that might be present are precluded by preliminary ester formation.
Remember, esters are weaker acylating reagents than either anhydrides or
acyl halides, as
noted earlier.
Finally, dicyclohexylcarbodiimide (DCC) effects the dehydration of a
carboxylic acid and amine mixture to the corresponding amide under
relatively mild conditions. The structure of this reagent and the mechanism
of its action have been
described. Its application to peptide synthesis will become apparent in
the following discussion.
The strategy for peptide synthesis, as outlined here, should now be apparent. The following example shows a selective synthesis of the dipeptide Ala-Gly.
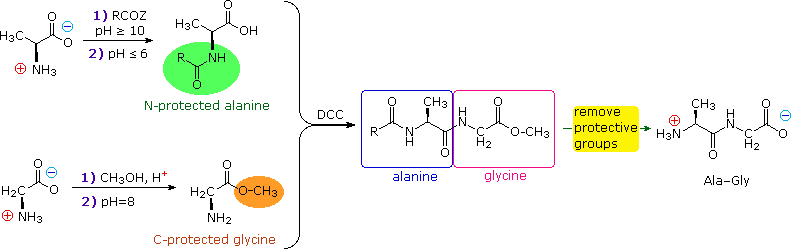
An important issue remains to be addressed. Since the N-protective group is an amide, removal of this function might require conditions that would also cleave the just formed peptide bond. Furthermore, the harsh conditions often required for amide hydrolysis might cause extensive racemization of the amino acids in the resulting peptide. This problem strikes at the heart of our strategy, so it is important to give careful thought to the design of specific N-protective groups. In particular, three qualities are desired:
1) The protective amide should be easy to attach to amino
acids.
2) The protected amino group should not react under peptide
forming conditions.
3) The protective amide group should be easy to remove under mild
conditions.
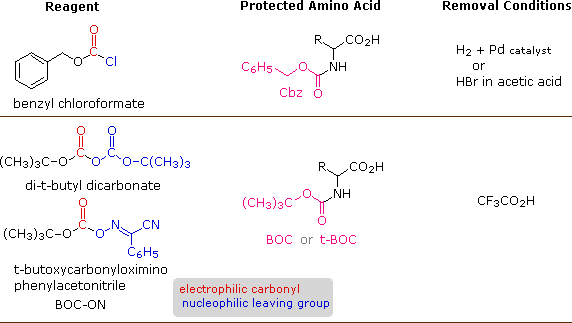
A number of protective groups that satisfy these conditions have been devised; and two of the most widely used, carbobenzoxy (Cbz) and t-butoxycarbonyl (BOC or t-BOC), are described here.
The reagents for introducing these N-protective groups are the acyl chlorides or anhydrides shown in the left portion of the above diagram. Reaction with a free amine function of an amino acid occurs rapidly to give the "protected" amino acid derivative shown in the center. This can then be used to form a peptide (amide) bond to a second amino acid. Once the desired peptide bond is created the protective group can be removed under relatively mild non-hydrolytic conditions. Equations showing the protective group removal will be displayed above by clicking on the diagram. Cleavage of the reactive benzyl or tert-butyl groups generates a common carbamic acid intermediate (HOCO-NHR) which spontaneously loses carbon dioxide, giving the corresponding amine. If the methyl ester at the C-terminus is left in place, this sequence of reactions may be repeated, using a different N-protected amino acid as the acylating reagent. Removal of the protective groups would then yield a specific tripeptide, determined by the nature of the reactants and order of the reactions.
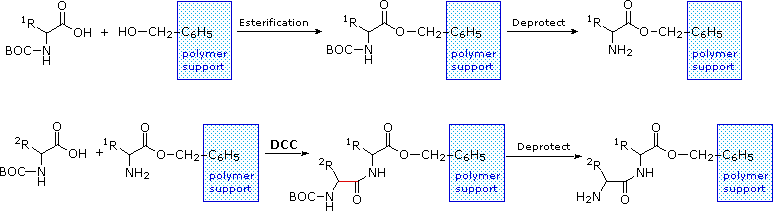
The synthesis of a peptide of significant length (e.g. ten residues) by this approach requires many steps, and the product must be carefully purified after each step to prevent unwanted cross-reactions. To facilitate the tedious and time consuming purifications, and reduce the material losses that occur in handling, a clever modification of this strategy has been developed. This procedure, known as the Merrifield Synthesis after its inventor R. Bruce Merrifield, involves attaching the C-terminus of the peptide chain to a polymeric solid, usually having the form of very small beads. Separation and purification is simply accomplished by filtering and washing the beads with appropriate solvents. The reagents for the next peptide bond addition are then added, and the purification steps repeated. The entire process can be automated, and peptide synthesis machines based on the Merrifield approach are commercially available. A series of equations illustrating the Merrifield synthesis may be viewed by clicking on the following diagram. The final step, in which the completed peptide is released from the polymer support, is a simple benzyl ester cleavage. This is not shown in the display.
The Merrifield Peptide Synthesis
Two or more moderately sized peptides can be joined together by selective peptide bond formation, provided side-chain functions are protected and do not interfere. In this manner good sized peptides and small proteins may be synthesized in the laboratory. However, even if chemists assemble the primary structure of a natural protein in this or any other fashion, it may not immediately adopt its native secondary, tertiary and quaternary structure. Many factors, such as pH, temperature and inorganic ion concentration influence the conformational coiling of peptide chains. Indeed, scientists are still trying to understand how and why these higher structures are established in living organisms.
Denaturation
The natural or native structures of proteins may be altered, and their
biological activity changed or destroyed by treatment that does not disrupt
the primary structure. This denaturation is often done deliberately
in the course of separating and purifying proteins. For example, many
soluble globular proteins precipitate if the pH of the solution is set at
the pI of the protein. Also, addition of trichloroacetic acid or the
bis-amide urea (NH2CONH2) is commonly used to effect
protein precipitation. Following denaturation, some proteins will return to
their native structures under proper conditions; but extreme conditions,
such as strong heating, usually cause irreversible change.
Some treatments known to denature proteins are listed in the following
table.
Denaturing Action |
Mechanism of Operation |
|---|---|
|
Heat |
hydrogen bonds are broken by increased translational and
vibrational energy. (coagulation of egg white albumin on frying.) |
|
Ultraviolet Radiation |
Similar to heat (sunburn) |
|
Strong Acids or Bases |
salt formation; disruption of hydrogen bonds. (skin blisters and burns, protein precipitation.) |
|
Urea Solution |
competition for hydrogen bonds. (precipitation of soluble proteins.) |
|
Some Organic Solvents |
change in dielectric constant and hydration of ionic groups. (disinfectant action and precipitation of protein.) |
|
Agitation |
shearing of hydrogen bonds. (beating egg white albumin into a meringue.) |
Not all proteins are easily denatured. As noted above, fibrous proteins such as keratins, collagens and elastins are
robust, relatively insoluble, quaternary structured proteins that play
important roles in the physical structure of organisms. Secondary
structures such as the α-helix and β-sheet take on a dominant role in the
architecture and aggregation of keratins. In addition to the intra- and
intermolecular hydrogen bonds of these structures, keratins have large
amounts of the sulfur-containing amino acid Cys, resulting in disulfide
bridges that confer additional strength and rigidity. The more flexible and
elastic keratins of hair have fewer interchain disulfide bridges than the
keratins in mammalian fingernails, hooves and claws. Keratins have a high
proportion of the smallest amino acid, Gly, as well as the next smallest,
Ala. In the case of β-sheets, Gly allows sterically-unhindered hydrogen
bonding between the amino and carboxyl groups of peptide bonds on adjacent
protein chains, facilitating their close alignment and strong binding.
Fibrous keratin chains then twist around each other to form helical
filaments.
Elastin, the connective tissue protein, also has a high percentage
of both glycine and alanine. An insoluble rubber-like protein, elastin
confers elasticity on tissues and organs. Elastin is a macromolecular
polymer formed from tropoelastin, its soluble precursor. The secondary
structure is roughly 30% β-sheets, 20% α-helices and 50% unordered. The
elastic properties of natural elastin are attributed to polypentapeptide
sequences (Val-Pro-Gly-Val-Gly) in a cross-linked network of randomly
coiled chains. Water is believed to act as a "plasticizer", assisting
elasticity.
Collagen is a major component of the extracellular
matrix that supports most tissues and gives cells structure. It has great
tensile strength, and is the main component of fascia, cartilage,
ligaments, tendons, bone and skin. Collagen contains more Gly (33%) and
proline derivatives (20 to 24%) than do other proteins, but very little
Cys. The primary structure of collagen has a frequent repetitive pattern,
Gly-Pro-X (where X is a hydroxyl bearing Pro or Lys). This kind of regular
repetition and high glycine content is found in only a few other fibrous
proteins, such as silk fibroin (75-80% Gly and Ala + 10% Ser). Collagen
chains are approximately 1000 units long, and assume an extended
left-handed helical conformation due to the influence of proline rings.
Three such chains are wound about each other with a right-handed twist
forming a rope-like superhelical quaternary structure, stabilized by
interchain hydrogen bonding.
Globular proteins are more soluble in aqueous
solutions, and are generally more sensitive to temperature and pH change
than are their fibrous counterparts; furthermore, they do not have the high
glycine content or the repetitious sequences of the fibrous proteins.
Globular proteins incorporate a variety of amino acids, many with large
side chains and reactive functional groups. The interactions of these
substituents, both polar and nonpolar, often causes the protein to fold
into spherical conformations which gives this class its name. In contrast
to the structural function played by the fibrous proteins, the globular
proteins are chemically reactive, serving as enzymes (catalysts), transport
agents and regulatory messengers.
 Although globular
proteins are generally sensitive to denaturation (structural unfolding),
some can be remarkably stable. One example is the small enzyme ribonuclease
A, which serves to digest RNA in our food by cleaving the ribose phosphate
bond. Ribonuclease A is remarkably stable. One procedure for purifying it
involves treatment with a hot sulfuric acid solution, which denatures and
partially decomposes most proteins other than ribonuclease A. This
stability reflects the fact that this enzyme functions in the inhospitable
environment of the digestive tract. Ribonuclease A was the first enzyme
synthesized by R. Bruce Merrifield, demonstrating
that biological molecules are simply chemical entities that may be
constructed artificially.
Although globular
proteins are generally sensitive to denaturation (structural unfolding),
some can be remarkably stable. One example is the small enzyme ribonuclease
A, which serves to digest RNA in our food by cleaving the ribose phosphate
bond. Ribonuclease A is remarkably stable. One procedure for purifying it
involves treatment with a hot sulfuric acid solution, which denatures and
partially decomposes most proteins other than ribonuclease A. This
stability reflects the fact that this enzyme functions in the inhospitable
environment of the digestive tract. Ribonuclease A was the first enzyme
synthesized by R. Bruce Merrifield, demonstrating
that biological molecules are simply chemical entities that may be
constructed artificially.
By clicking the cartoon image on the left, an interactive model of
ribonuclease A will be displayed.