Identifying Polar Covalent Bonds
Examine structural formulas A through L. Identify those covalent bonds within each structure that would be classified as polar. For this problem we shall assume that C-C and C-H bonds are not polar, because the electronegativity differences are small. The questions posed below require you to enter appropriate letters in the answer box for each question. Do not enter superfluous characters, since they will be counted as incorrect answers.
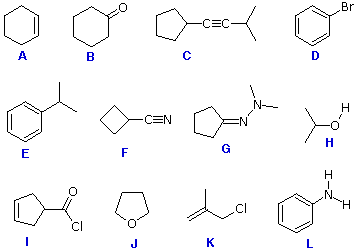
The "View Answer" part of this problem does not function using Internet Explorer ver.3 and earlier.