Distinguishing Isomers
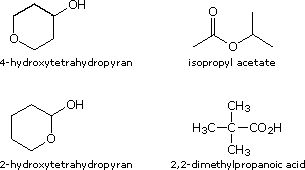 Four isomeric
(C5H10O2) liquids are found in separate
bottles with no labels. We shall refer to these unknowns as A, B, C, & D. These
compounds are known to have the structures shown on the right. Each of
these compounds is partially or completely soluble in water, and is also
soluble in ether and methylene chloride.
Four isomeric
(C5H10O2) liquids are found in separate
bottles with no labels. We shall refer to these unknowns as A, B, C, & D. These
compounds are known to have the structures shown on the right. Each of
these compounds is partially or completely soluble in water, and is also
soluble in ether and methylene chloride.
To discover which is which, three chemical tests may be
applied to these unknowns:
1. Treatment with 5% aqueous sodium bicarbonate
(NaHCO3 ).
2. Treatment with Tollens' reagent (AgNO3 in
NH4OH ) .
3. Treatment with Jones' reagent (CrO3 in
H3O(+) ).
|
|
|---|
Once you have conducted your tests, make structural assignments to A, B, C & D in the boxes following the compound names.
| 4-Hydroxytetrahydropyran .... 2-Hydroxytetrahydropyran .... Isopropyl Acetate .................... 2,2-Dimethylpropanoic acid ... |