UV-Visible Spectroscopy
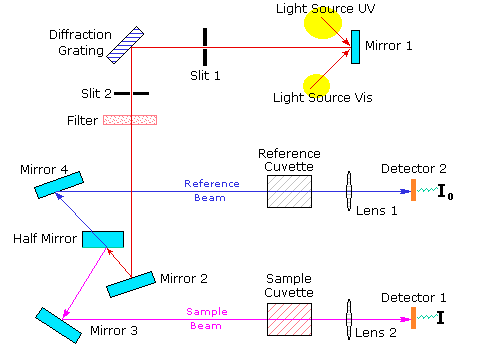
A diagram of the components of a typical spectrometer are shown in the following diagram. The functioning of this instrument is relatively straightforward. A beam of light from a visible and/or UV light source (colored red) is separated into its component wavelengths by a prism or diffraction grating. Each monochromatic (single wavelength) beam in turn is split into two equal intensity beams by a half-mirrored device. One beam, the sample beam (colored magenta), passes through a small transparent container (cuvette) containing a solution of the compound being studied in a transparent solvent. The other beam, the reference (colored blue), passes through an identical cuvette containing only the solvent. The intensities of these light beams are then measured by electronic detectors and compared. The intensity of the reference beam, which should have suffered little or no light absorption, is defined as I0. The intensity of the sample beam is defined as I. Over a short period of time, the spectrometer automatically scans all the component wavelengths in the manner described. The ultraviolet (UV) region scanned is normally from 200 to 400 nm, and the visible portion is from 400 to 800 nm.
|
|
||||
|---|---|---|---|---|
|
If the sample compound does not absorb light of of a given
wavelength, I = I0. However, if the sample
compound absorbs light then I is less than
I0, and this difference may be plotted on a graph
versus wavelength, as shown on the right. Absorption may be
presented as transmittance (T =
I/I0) or absorbance
(A= log I0/I). If no
absorption has occurred, T = 1.0 and A= 0. Most
spectrometers display absorbance on the vertical axis, and the
commonly observed range is from 0 (100% transmittance) to 2 (1%
transmittance). The wavelength of maximum absorbance is a
characteristic value, designated as λmax.
|
|
|||
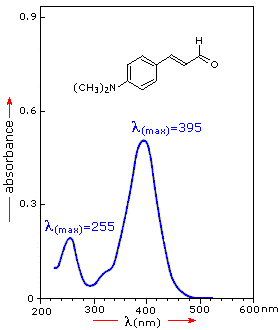
For the spectrum on the right, a solution of 0.249 mg of the unsaturated
aldehyde in 95% ethanol (1.42 • 10-5 M) was placed in a 1
cm cuvette for measurement. Using the above formula, ε = 36,600 for the 395
nm peak, and 14,000 for the 255 nm peak. Note that the absorption extends
into the visible region of the spectrum, so it is not surprising that this
compound is orange colored.
Molar absorptivities may be very large for strongly
absorbing compounds (ε >10,000) and very small if absorption is weak (ε
= 10 to 100).
End of this supplementary topic
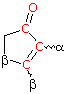
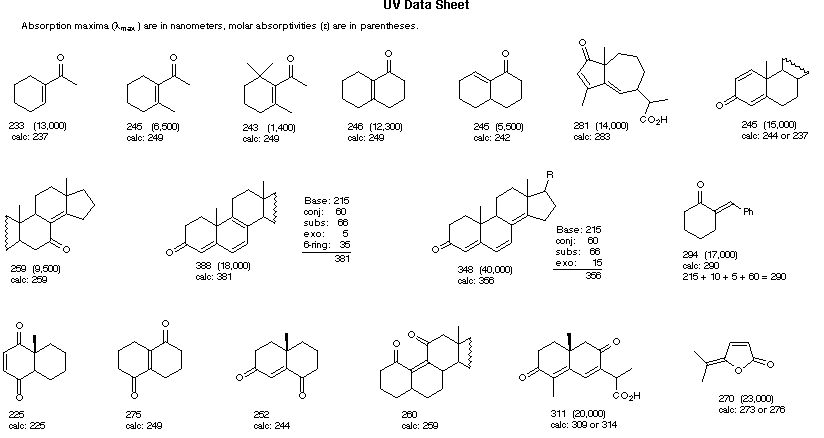
Empirical Rules for Absorption Wavelengths of Conjugated Systems
Woodward-Fieser Rules for Calculating the λmax of Conjugated Dienes and Polyenes
|
Core Chromophore |
Substituent and Influence |
|---|---|
|
|
R- (Alkyl Group) .... +5 nm RO- (Alkoxy Group) .. +6 X- (Cl- or Br-) ......... +10 RCO2- (Acyl Group) .... 0 RS- (Sulfide Group) .. +30 R2N- (Amino Group) .. +60 C6H5 (Phenyl Group) ... +60 |
 Cyclohexadiene* 260 nm |
|
|
|
|
λmax (calculated) = Base (215 or 260) +
Substituent Contributions
Some examples that illustrate these rules follow.
Woodward-Fieser Rules for Calculating the π __> π* λmax of Conjugated Carbonyl Compounds
|
Core Chromophore |
Substituent and Influence |
|---|---|
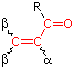 R = Alkyl 215
nm R = Alkyl 215
nmR = H 210 nm R = OR' 195 nm |
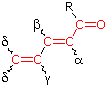
α- Substituent C6H5 (Phenyl Group) ... +60 |
 Cyclopentenone 202 nm |
|
 |
|
|
|
|
λmax (calculated) = Base + Substituent Contributions and Corrections
Some examples that illustrate these rules follow.
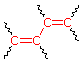
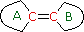 (i) Each
exocyclic double bond adds 5 nm. In the example on the
right, there are two exo-double bond components: one to ring A
and the other to ring B.
(i) Each
exocyclic double bond adds 5 nm. In the example on the
right, there are two exo-double bond components: one to ring A
and the other to ring B.