Stereochemical Terminology
The asymmetric carbon concept has played an important role in the
development of organic chemistry. Indeed, it provided the focus for the
revolutionary structural theory introduced by Jacobus van't
Hoff and Joseph A. Le Bel. However, in recent years the nuance of
distinguishing between the purely structural definition of an asymmetric
carbon and the operational definition of a stereogenic element (e.g. a
chiral center) has become increasingly important. 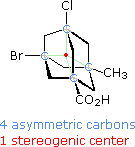 For example, consider the
tetrasubstituted adamantane molecule shown on the left. Although four
asymmetric carbon atoms (colored light blue) are present in this compound,
it exists in the form of only two stereoisomers (enantiomers) rather than
the sixteen predicted by the 2n rule. There is, in fact, only
one stereogenic center, shown by the red dot at the center of the molecule,
and the configuration shown here is (R). The asymmetric carbon atoms
are not stereogenic centers, because any two substituents on any of these
bridgehead carbons cannot be exchanged without destroying the
constitutional integrity of this rigid, highly bridged molecule.
For example, consider the
tetrasubstituted adamantane molecule shown on the left. Although four
asymmetric carbon atoms (colored light blue) are present in this compound,
it exists in the form of only two stereoisomers (enantiomers) rather than
the sixteen predicted by the 2n rule. There is, in fact, only
one stereogenic center, shown by the red dot at the center of the molecule,
and the configuration shown here is (R). The asymmetric carbon atoms
are not stereogenic centers, because any two substituents on any of these
bridgehead carbons cannot be exchanged without destroying the
constitutional integrity of this rigid, highly bridged molecule.
The configurations of the asymmetric bridgehead carbon units in this
structure are (R), (S), (R) and (R)
respectively, for the Br, Cl, CO2H and CH3
substituents. Its enantiomer would. of course, have the opposite
configurations.
End of this supplementary topic |
|---|
Oxygen Molecular Orbital Diagram
Simple covalent bond models involving electron pair
sharing have proven to be a valuable tool for describing the structures
of simple molecules, such as methane, water, fluorene and nitrogen.
However, this model fails to explain the paramagnetic nature of oxygen. The
electrons in most non-metallic compounds are paired (opposite spins) in
bonding and non-bonding orbitals, resulting in a zero spin diamagnetic
molecule. However, oxygen has a net electron spin of 1, indicating there
are two unpaired electrons having the same spin (1/2). One of the triumphs
of molecular orbital theory has been its success in rationalizing this
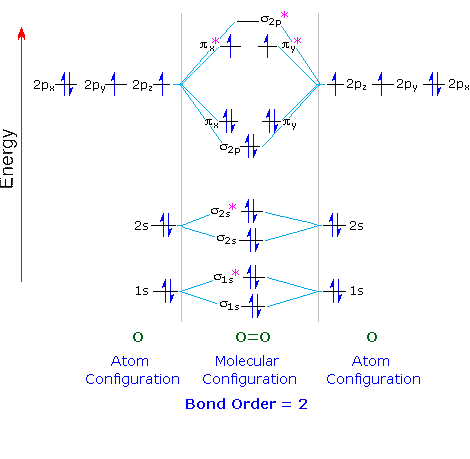
fact, as shown in the following correlation diagram.
Each oxygen atom contributes eight electrons to the molecule. Half of these
occupy sigma bonding and antibonding orbitals, formed by overlap of 1s and
2s orbitals, that do not contribute to the final bonding picture. Of the
remaining eight electrons, six occupy 2σ and 2π bonding orbitals in accord
with the aufbau principle. This leaves two electrons which must be placed
in the two 2π* antibonding orbitals. By Hund's Rule,
these electrons will each occupy one of these degenerate (same energy)
orbitals, and have identical spins. The resulting antibonding character
cancels one of the three bonding interactions, leaving a total bond order
of 2.
| Correlation diagrams for fluorine and nitrogen will be displayed above by clicking the appropriate button. | ||||
End of this supplementary topic |
|---|
Unusual Fatty Acids
A wide array of unusual fatty acids is found in seed oils and
microorganisms such as bacteria. Among these are fatty acids containing
cis-cyclopropane fatty acids (e.g. lactobacillic acid), omega-cycloalkyl
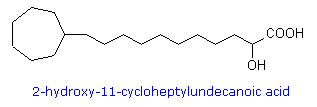
fatty acids (e.g. 2-hydroxy-11-cycloheptylundecanoic acid) and cyclopropene
fatty acids (e.g. malvalic and sterculic acid).
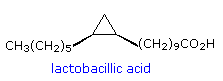 |
 |
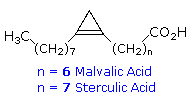 |
|---|
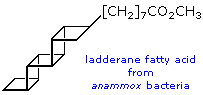
Lactobacillic acid has been found in a wide range of bacterial species, both gram-negative and gram-positive, ranging from strict anaerobes to aerobes. It is often accompanied by cis-9,10-methylene-hexadecanoic acid, and other homologues (C14 to C20 in chain-length). Some organisms contain cis-9,10-methylene-octadecanoic acid (dihydrosterculic acid), the cyclopropane analogue of oleic acid, together with homologous fatty acids (C16 or C20 in chain-length). In the bacterium Alicyclobacillus cycloheptanicus, 11-Cycloheptylundecanoic, the 2-hydroxy analogue, and 13-cycloheptyltridecanoic acids, together with three minor homologues, comprise nearly 80% of the fatty acids of this species. In Litchi chinensis seed oil, dihydrosterculic acid is the major carbocyclic fatty acid, whereas Sterculia foetida seed oil contains 65-78% cyclopropene lipids, principally sterculic acid.
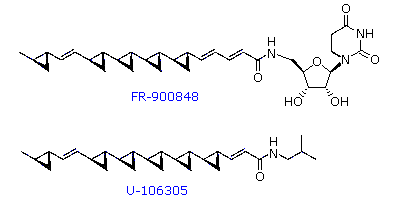 Two novel
polycyclopropane fatty acid derivatives, FR-900848 and U-106305, were
isolated from Streptoverticillium fervens by Japanese
microbiologists in the early 1990's. Structural formulas for these
remarkable compounds are drawn on the right. FR-900848 shows potent,
selective activity against filamentous fungi such as Aspergillus niger,
Mucor rouxianus, Aureobasidium pullulans, and various Trichophyton
sp. U-106305 is an inhibitor of cholesteryl ester transfer protein
(CETP).
Two novel
polycyclopropane fatty acid derivatives, FR-900848 and U-106305, were
isolated from Streptoverticillium fervens by Japanese
microbiologists in the early 1990's. Structural formulas for these
remarkable compounds are drawn on the right. FR-900848 shows potent,
selective activity against filamentous fungi such as Aspergillus niger,
Mucor rouxianus, Aureobasidium pullulans, and various Trichophyton
sp. U-106305 is an inhibitor of cholesteryl ester transfer protein
(CETP).
A few years after their discovery, these compounds were synthesized by
British and American chemists. Biosynthetic studies indicate that the
carbon backbone is derived from acetate via the polyketide pathway typical
of fatty acid biosynthesis. The methylene carbon of each cyclopropane then
originates from the methyl group of methionine. Thus, a polyunsaturated
fatty acid, produced by the usual polyketide pathway, appears to be
cyclopropanated through the sequential addition of methylene groups by a
separate methyltransferase enzyme.
In the late 1980's microbiologists in the Netherlands discovered a remarkable red-colored bacterium that anaerobically oxidized ammonia to elemental nitrogen. Although a simple overall conversion can be written as:
| NH4+ + NO2– |  |
N2 + 2H2O |
|---|
The nitrogen cycle converts elemental nitrogen into more usable forms
(such as ammonia and nitrate ions) and back again, maintaining a global
balance, as shown on the right. First, nitrogen gas is converted directly
to ammonia by nitrogen-fixing microbes. 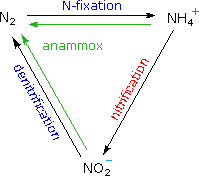 Plants and animals consume
ammonia, and then release it when they die and decompose. The next step in
the cycle is carried out by nitrifying bacteria and archaea, which
transform ammonia to nitrites and nitrates. The cycle is completed as
denitrifying microorganisms convert nitrates into nitrogen gas,
replenishing the atmosphere. Anammox bacteria, such as Brocadia
anammoxidans, Kuenenia stuttgartiensis and Scalindua sorokinii,
take a short cut through the cycle, creating a path from ammonia and
nitrite directly to nitrogen gas. Indeed, anammox bacteria in marine
ecosystems actively contribute to biological nitrogen cycling, being
responsible for at least 50% of total nitrogen production in the
oceans.
Plants and animals consume
ammonia, and then release it when they die and decompose. The next step in
the cycle is carried out by nitrifying bacteria and archaea, which
transform ammonia to nitrites and nitrates. The cycle is completed as
denitrifying microorganisms convert nitrates into nitrogen gas,
replenishing the atmosphere. Anammox bacteria, such as Brocadia
anammoxidans, Kuenenia stuttgartiensis and Scalindua sorokinii,
take a short cut through the cycle, creating a path from ammonia and
nitrite directly to nitrogen gas. Indeed, anammox bacteria in marine
ecosystems actively contribute to biological nitrogen cycling, being
responsible for at least 50% of total nitrogen production in the
oceans.
A practical use of anammox bacteria lies in wastewater treatment. Sewage
plants and industrial processes, such as fertilizer manufacture and
petroleum refining, generate millions of liters of ammonia-rich waste, all
of which needs to be broken down. Conventional methods use nitrifying
bacteria to convert ammonia into nitrite and nitrate, and then denitrifying
bacteria to yield nitrogen gas. The nitrifying microbes need oxygen, so
machines are needed to aerate the sludge. Also, the denitrifying bacteria
need an energy source, which they "burn" to produce carbon dioxide. This
fuel is often supplied in the form of methanol. The process is costly,
takes up a lot of space and produces a greenhouse gas. Anammox bacteria use
ammonia as their fuel, hence there's no need for carbon fuels such as
methanol. They do not need oxygen, so the process uses less electricity,
and instead of producing carbon dioxide, anammox bacteria consume it, so
the method is environmentally friendly. Altogether, this leads to a 90%
reduction in operational costs and a 50% reduction in space, compared with
conventional methods.
End of this supplementary topic |
|---|